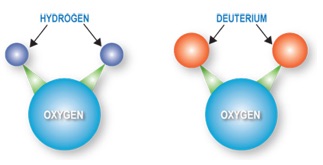
Deuterium oxide, also known as “heavy water” or “deuterium water”, is the compound of oxygen and the heavy isotope of hydrogen, called deuterium. Physically and chemically, heavy water is almost identical to ordinary “light” water, H₂O. It is called heavy water because its density is greater than H₂O. Its chemical formula is D₂O.
Deuterium contains one neutron and one proton in its nucleus, which makes it twice as heavy as protium (hydrogen), which contains only one proton. Deuterium oxide is a colorless and odorless liquid at normal temperature and pressure. Compared to ordinary water, its chemical characteristic is relatively inactive with a specific gravity of 1.10775 (at 25℃), melting/freezing point of 3.82℃, and a boiling point of 101.42℃. The hydrogen bond strength and degree of association between heavy water molecules are both stronger than that of ordinary water molecules.